- Home
- About
- Contact
- Swiper carousel unpkg
- Spintiers mudrunner mod
- 6 month old black baby girl hairstyles
- Bakery story 2 forum
- Knicks julius randle wife
- Dragon spring phoenix rise nyc the shed
- Dlc quest walkthrough
- Play vlc streamer iphone to tv
- Braces brackets
- Dynamic light scattering size distribution by number
- Super eraser code
- Transloader rental seguin texas
- Pdf merger
- Audio enhancer bongiovi dps plugi
- Goldenkey hack borderlands 2
- Ubuntu soundconverter
- Cnet winzip for mac
- Professional name for busboy

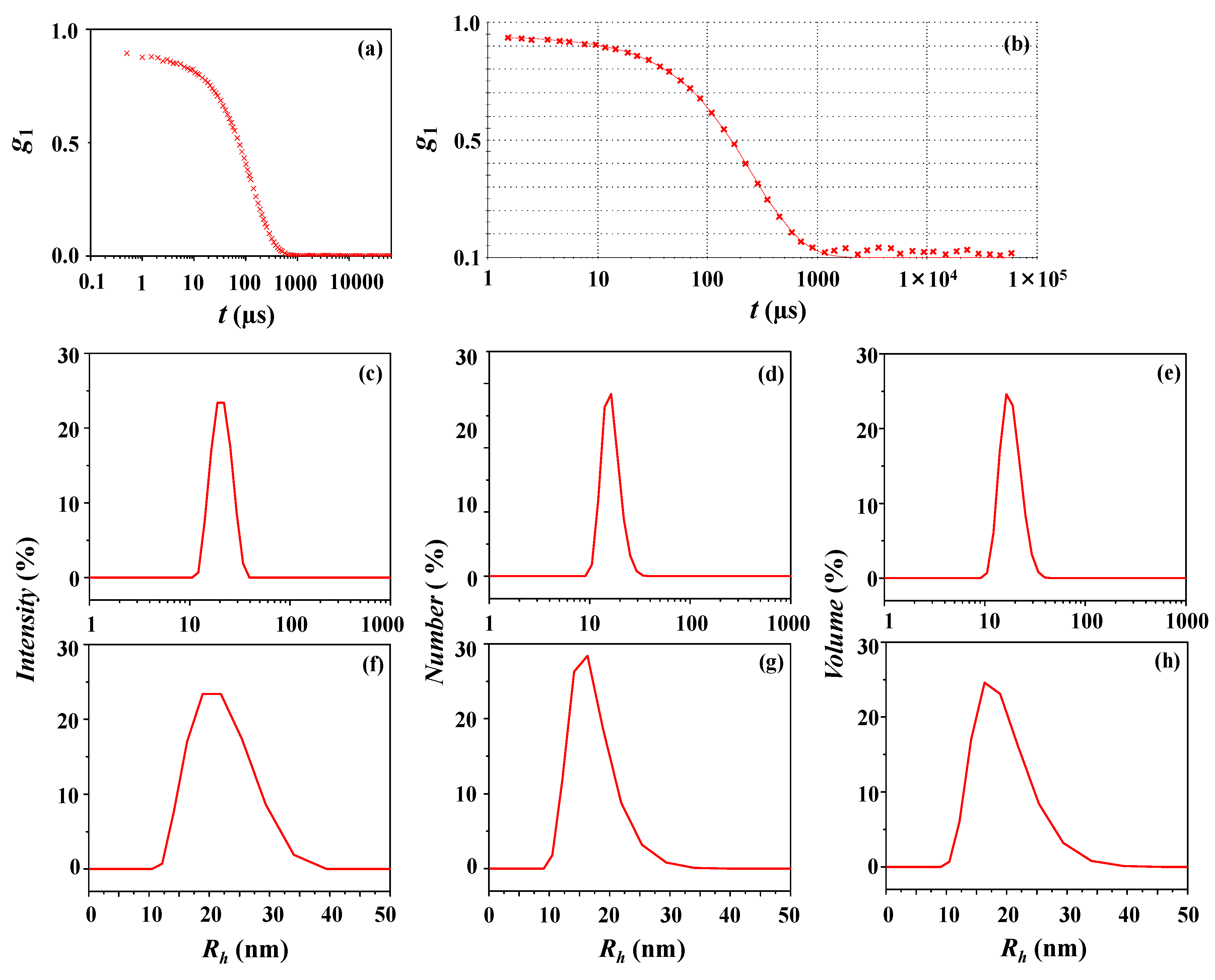
In 2003, a bulk OD method to determine AAV capsid (Cp) and vector genome (Vg) titer was proposed as an alternative to qPCR and ELISA. Despite their historic and continued use, each method has its own limitations, such as low throughput or high variability 7, 8.īulk optical density (OD) measurements have been used to quantify the vector genome and capsid protein content of adenovirus preparations 9, 10. Various methods, including electron microscopy, dynamic light scattering, analytical ultracentrifugation, ELISA, and PCR are used to independently monitor purified AAV size, aggregation propensity, stability, empty-to-full capsid ratio, capsid (Cp) and vector genome (Vg) titer, respectively. Therefore, a successful gene therapy program depends on accurate vector characterization and titration, which dictates the safety and efficacy in humans. Previous gene therapy programs have shown positive correlation in general between the presence of gene copy numbers (vg/kg patient weight) and protein expression 5, 6. While several successful methods exist for large-scale, clinical AAV production 2, 3, 4, the development of robust analytics to assess variables in the overall process development and final AAV drug products is an iterative process. As of April 2020, 244 gene therapy trials using AAVs are ongoing worldwide, 24 of which are in Phase III clinical trials, ( ). Due to their relative safety and long-term gene expression, multiple serotypes of recombinant AAV vectors are in use across gene therapy programs at the clinical and research stage 1. AAV particles consist of a small ~ 25 nm icosahedral capsid composed of three types of structural proteins (VP1, VP2, and VP3) coating a single-stranded ~ 4.7 kb genome. These novel applications designate this well-established method as a powerful tool for product development and process analytics in future gene therapy programs.Īdeno-associated virus (AAV) belongs to the genus Dependoparvovirus in the family Parvoviridae.
DYNAMIC LIGHT SCATTERING SIZE DISTRIBUTION BY NUMBER SERIES
In the current study, a new series of equations for SEC-MALS are used in tandem with intrinsic properties of the capsids and encapsidated DNA to quantify multiple physical AAV attributes in one 20-min run with minimal sample manipulation, high accuracy, and high precision. Currently, multiple techniques are required to generate this information, with varying accuracy and precision. The current study demonstrates that this method provides detailed AAV characterization information, including but not limited to aggregation profile, size-distribution, capsid content, capsid molar mass, encapsidated DNA molar mass, and total capsid and vector genome titer. Size exclusion chromatography (SEC) coupled to multiangle light scattering (MALS) offers a straightforward approach to comprehensively characterize AAV capsids. Complete and precise characterization of capsid particles, including capsid and vector genome concentration, is necessary to safely and efficaciously dose patients. Adeno associated virus (AAV) capsids are a leading modality for in vivo gene delivery.

